Phase separation is considered a crucial process in disease progression, since proteins involved in amyloid-associated pathologies, like FUS, play also a crucial role in phase separation. FUS contains, like other phase separating proteins, a disordered domain. The marginal stability and/or disorder of this domain is crucial to respond to environmental and cellular change, but these properties also dispose the proteins sensitive to misfolding and aggregation, in particular under stress.
Thus, biomolecular condensates are closely connected to misfolding and aggregation and amyloid-associated pathologies. An example of condensates is the stress granule (SG), which copes with stresses such as heat stress (HS). SGs have diverse functions including the stimulation of the immune response, modulation of signaling pathways or by serving as PQC compartments, transiently sequestering, and storing unfolded or misfolded proteins.
A protein that was identified to accumulate inside SGs comprises superoxide dismutase 1 (SOD1). SOD1 is an essential enzyme that catalyzes the clearance of radical oxygen species and matter of continuous intensive research due to the intricate link between its folding and aggregation properties and amyotrophic lateral sclerosis (ALS) pathology. If the accumulation of unfolded, misfolded, or aggregated SOD1 inside SGs promotes or endangers protein homeostasis and cell viability is so far unknown.
In the first funding period of SPP2191, we found that destabilized mutants of SOD1bar preferentially accumulate inside SGs. However, reconstituted in vitro experiments suggested that the increased flexibility and hydrophobicity of the unfolded state is only a minor driving force for partitioning. Crowding experiments revealed that partitioning depends on specific protein-protein interactions and valence, depending on site-specific mutations as well as on the chemical nature of the crowder.
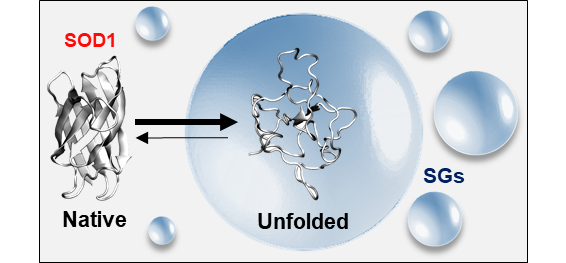
Unfolded state of destabilised ALS-related SOD1 mutant accumulates inside SGs
Aim of the project:
We would like to extend our research to measure SOD1 unfolding, misfolding, and self-association processes in SGs in direct comparison to the mutual processes occurring in the cytoplasm. The project will significantly contribute to establish the so far missing link between protein folding and aggregation pathways and condensate formation. We aim to investigate how SGs reshape folding and aggregation pathways to assist protein homeostasis.
A current view is that sequestration may lead to a removal of unstable and possibly toxic species from the cytoplasm, ‘storing’ or ‘buffering’ them in condensates for degradation, therefore possibly shielding them from further homotypic aggregation. This mechanism would support cytoplasmic PQC machinery and thereby ensure protein homeostasis. On the other hand, if the ‘buffering’ capacity is overloaded, e.g. under prolonged stresses, the sequestration can be harmful to condensate integrity leading to aberrant liquid-to-solid phase transitions. This current knowledge is mainly derived from measuring the enrichment of disease-related proteins in condensates paired with their ability to induce aberrant phase transitions. Our research aims to advance this knowledge by a consideration of the protein’s conformational states and dynamics along its folding and aggregation pathway.
The key questions are:
- Are folded, unfolded, misfolded, oligomeric, aggregated, or fibrillar states preferentially sequestered by condensates?
- Which of the steps along the aggregation pathway is inhibited, accelerated, or redirected to the condensate environment?
- Are aggregates mainly nucleated in the cytoplasm or within condensates and how does the subsequent maturation of aggregates differ in these environments?
We address these questions by thermodynamic and kinetic experiments capable of directly comparing mutual folding and aggregation pathways in the cytoplasm and SGs of single living cells. We investigate different SOD1 variants and mutants and conduct the experiments under different conditions to elaborate the role of cellular factors such as molecular chaperones. The emerging model will allow us to define the active or passive role of condensates in regulating aggregation pathways under cell stress.
The Team:

Simon Ebbinghaus
Dr.rer.nat
Simon Ebbinghaus received his doctoral degree (Dr rer. nat.) from the Ruhr-University Bochum in 2007 under the supervision of Martina Havenith and worked as a Feodor Lyne Research Fellow with Martin Gruebele at the University of Illinois (Urbana-Champaign) from 2008 to 2010. He was appointed as an Assistant Professor at the Ruhr-University Bochum in 2011 and Associate Professor in 2017. In April 2018 he moved to the TU Braunschweig as a Full Professor.
Technische Universität Braunschweig
BRICS / Institut für Physikalische und Theoretische Chemie
Rebenring 56
38106 Braunschweig

Mailin Becker
Sara Ribeiro
Erik Schernich
Related Publications
Samanta, N. et al. (2021) Sequestration of Proteins in Stress Granules Relies on the In-Cell but Not the In Vitro Folding Stability. J. Am. Chem. Soc. 143, 19909–19918
Hautke, A.C. and Ebbinghaus, S. (2021) Folding Stability and Self-Association of a Triplet-Repeat (CAG)20 RNA Hairpin in Cytomimetic Media. ChemSystemsChem 3, e2000052
Ribeiro, S.S. et al. (2019) The synergic effect of water and biomolecules in intracellular phase separation. Nat Rev Chem 3, 552–561
Ribeiro, S. et al. (2018) Protein folding and quinary interactions: creating cellular organisation through functional disorder. FEBS Letters 592, 3040–3053

